 Which elements are the less reactive in the periodic table? Bromine, Iodine and sulfur, for another 3. The most reactive nonmetals would be fluorine, oxygen, and chlorine, just to name 3. What are three of the most reactive nonmetals? So, they are the least reactive non-metals. They are considered as least reactive elements and as they are categorized as non-metals. Group VIII-A elements have fully filled orbitals and hence are most stable amongst all the elements in the periodic table. Which group of nonmetals is the least reactive? As a result, it oxidizes all other substances. Due to its strong electrical negativity and small size, fluorine has a strong tendency to accept electrons from other atoms or ions. What elements are the most reactive of all nonmetals?įluorine is the most reactive non-metal because it is the most electrolytic of all non-metallic elements in the periodic table. They are highly reactive nonmetals with seven valence electrons. Which group is the most reactive nonmetals?Įlements in group 17 are called halogens (see Figure below). 
Rest of the nonmetals are solids at room temperature. Bromine (Br) is a nonmetal which is in liquid state at room temperature. 11 non metals are in gaseous state at room temperature. Nonmetals are found in solid state, liquid state as well as gaseous state. By obtaining an electron, halogens form anions with a -1 charge. They have 7 electrons in their outermost shell and desire to gain an extra electron to complete their shell of 8 electrons. The halogens are so reactive due to their electronic configuration. Halogens are the most reactive nonmetals on the periodic table. Which is the most reactive group of nonmetals *? 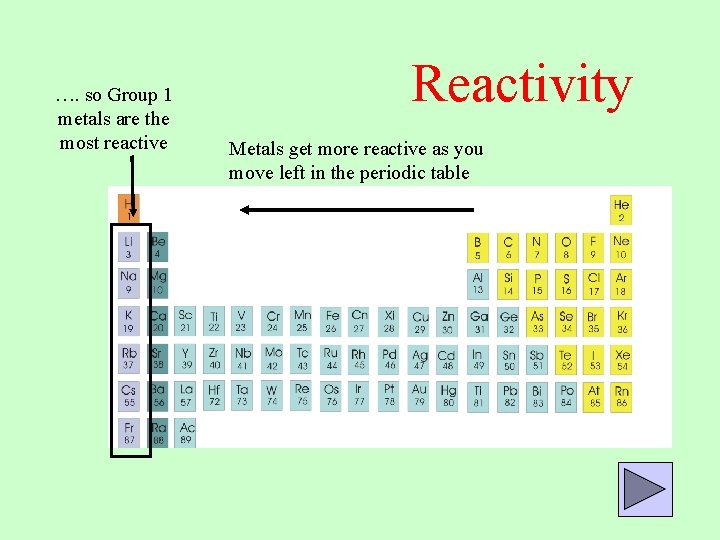
In the nonmetals, reactivity increases as you move up a group, and to the left. The trend in reactivity in the nonmetals is the opposite of the trend in the metals. The most reactive nonmetal is fluorine, F. 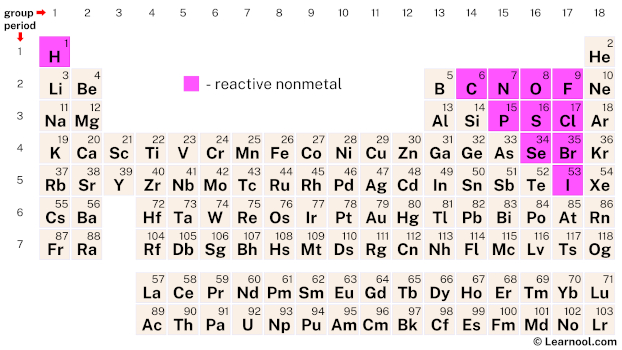
Where do you find the most reactive nonmetals on the periodic table?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
